Plastická chirurgie Péče o zdraví - hradí pojišťovna Péče o krásu - hradí pacient
Gynekologie Prenatální diagnostika
Lékařská genetika Urologie RTG Ortopedie Rehabilitace
Ordinační doba Kariéra Restaurace Blog
Nevíte si rady? Kontaktujte nás
prostřednictvím našeho formuláře
zde.



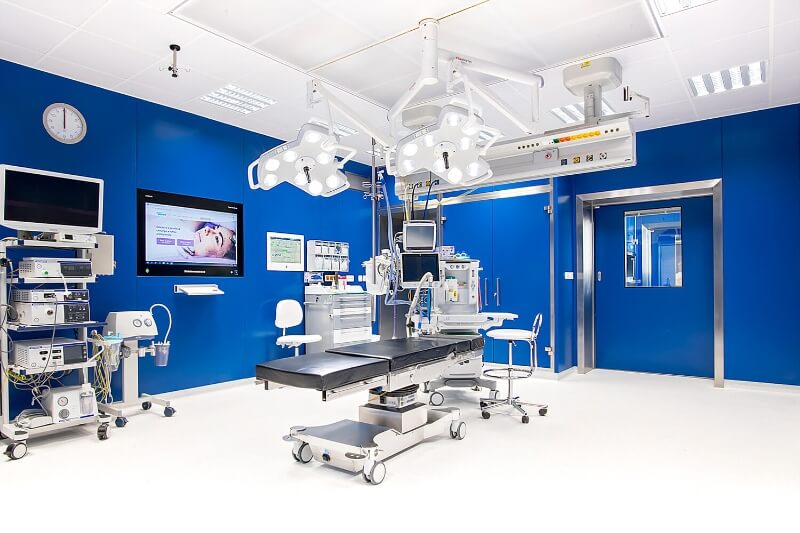
Kvalitní lékařská
péče pro celou
rodinu
Provoz o Velikonocích
Provoz o Velikonocích bude na klinikách Sanus omezen. Ambulance budou v pátek 29.3. a v pondělí 1.4. uzavřeny…
Valentýnský dárek pro klienty centra asistované reprodukce
Valentýnský dárek, který trvá navždy. Udělejte první krok v novém roce k vytouženému miminku ❤️
Proč SANUS?
Budete v dobrých rukou skutečných profesionálů ve svých oborech.
Klademe důraz na osobní přístup a spokojenost klienta. Hospitalizační péče probíhá v příjemném prostředí soukromé nemocnice s charakterem sanatoria. Většina námi poskytované zdravotní péče je hrazena z veřejného zdravotního pojištění. Děláme moderní medicínu 21. století. Klientům se snažíme nabídnout co nejkratší objednací lhůty. Naše centra asistované reprodukce se nachází ve 3 krajských
městech – Hradec Králové, Jihlava a Pardubice.

28 let zkušeností

Komplexnost
a dostupnost péče 24/7

Více než 4 000 zákroků
v celkové anestezii ročně

65 000 návštěv klientů
našich ambulancí ročně

1 000 léčebných cyklů
asistované reprodukce ročně
Více než 10 000 narozených dětí

Pracuje pro nás
více než 250 odborníků
Novinky
PROVOZ O VELIKONOCÍCH
Provoz o Velikonocích bude na klinikách Sanus omezen. Ambulance budou v pátek 29.3. a v pondělí 1.4. uzavřeny…
VALENTÝNSKÝ DÁREK PRO KLIENTY CENTRA ASISTOVANÉ REPRODUKCE
Valentýnský dárek, který trvá navždy. Udělejte první krok v novém roce k vytouženému miminku ❤️
REKONSTRUKCE V ROCE 2023
Poslední prosincový týden roku 2023 proběhla rekonstrukce vyšetřoven na urologii a gynekologii v Sanus Hradec…
NOVOROČNÍ DÁREK PRO KLIENTY CENTRA ASISTOVANÉ REPRODUKCE
Rezervujte si svůj první termín lednové konzultace do 31.01.2024 a získejte dárek v hodnotě 3000 Kč na…
Centra asistované reprodukce SANUS se nachází ve 3 krajích
Hradec Králové
Pardubice
Jihlava
Sanatorium SANUS
Labská kotlina 1220/69, 500 02 Hradec Králové

Centrum asistované reprodukce
Rokycanova 2798, 530 02 Pardubice

Centrum asistované reprodukce
Vrchlického 59, 586 33 Jihlava

Spolupracujeme s pojišťovnami